 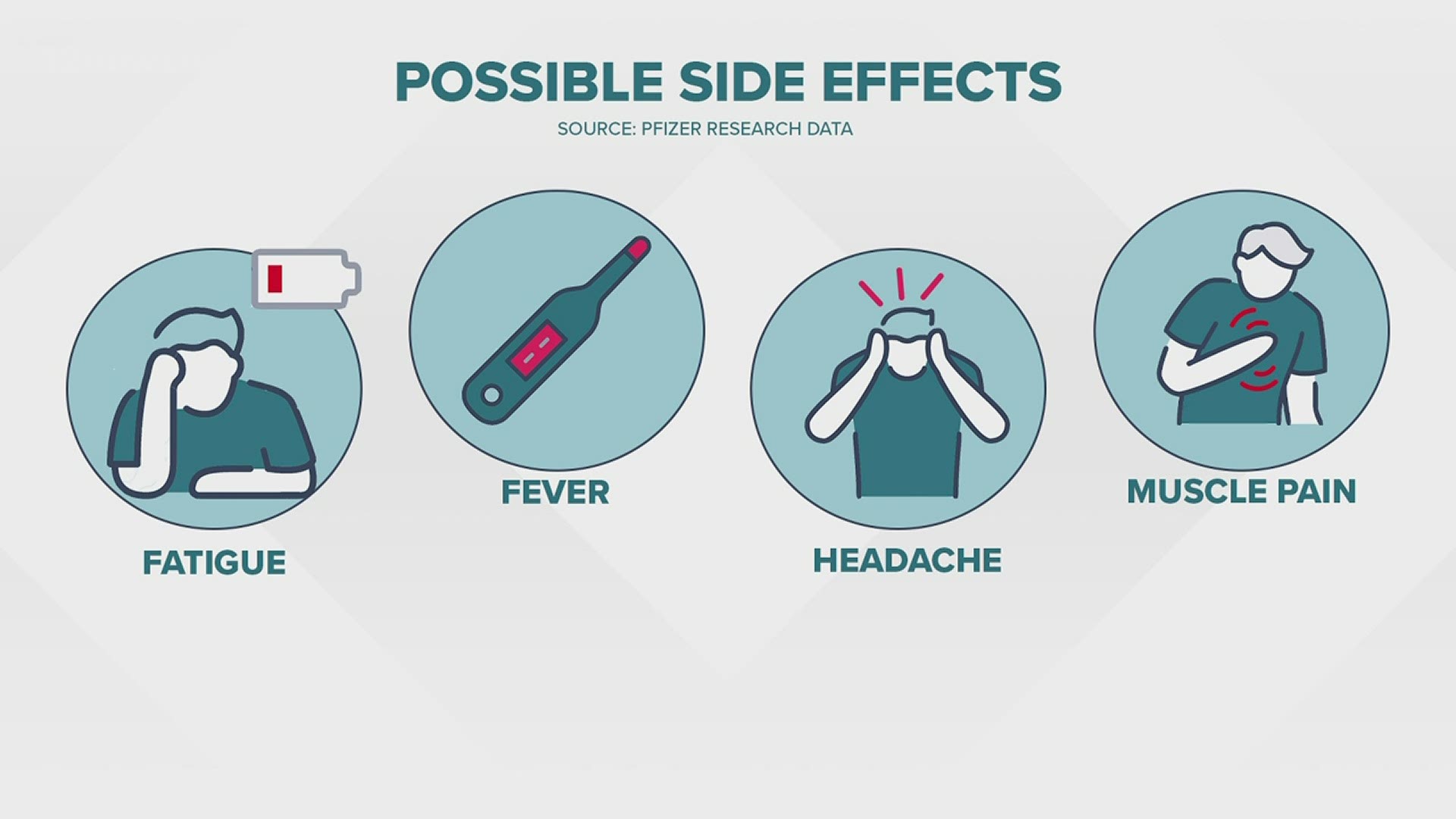
51% effective in trial participants 6 to 23 months old.37% effective in trial participants 2 to 5 years old.as effective in trial participants 6 to 11 years old as in young adults (18 to 25 years old).100% effective in trial participants 12 to 17 years old.94.1% effective in protecting trial participants aged 18 and above against COVID-19.EffectivenessĬlinical trials showed that beginning 2 weeks after the second dose, the Moderna Spikevax ® COVID vaccine was: Its safety and effectiveness in younger people has not yet been established.  
The Moderna Spikevax ® Bivalent COVID-19 vaccine (Original/Omicron BA.4/5) is approved as a booster for people who are 6 years of age and older. Moderna Spikevax ® Bivalent COVID-19 vaccine (Original/Omicron BA.4/5) The Moderna Spikevax ® Bivalent COVID-19 vaccine (Original/Omicron BA.1) is approved as a booster for people who are 6 years of age and older. Moderna Spikevax ® Bivalent COVID-19 vaccine (Original/Omicron BA.1) This vaccine is also approved as a booster for people 12 years of age and older. Its safety and effectiveness in people younger than 6 months have not yet been established. The Moderna Spikevax ® COVID-19 vaccine is approved for primary vaccination in people who are 6 months of age and older. Who the vaccines are approved for Moderna Spikevax ® COVID-19 vaccine Vaccine review, approval and monitoring.Number of doses: 2 doses for primary series and 1 dose as a booster On this page How it's given: Injection in muscle (usually the upper arm) KFF COVID-19 vaccine monitor: April 2021.Approved for: Primary series in individuals age 6 months and older, or as a booster dose in individuals age 12 years and older (Moderna Spikevax ® COVID-19 vaccine)Īs a booster dose in individuals age 6 years and older (Moderna Spikevax ® Bivalent COVID-19 vaccine (Original/Omicron BA.1)Īs a booster dose in individuals age 6 years and older (Moderna Spikevax ® Bivalent COVID-19 vaccine (Original/Omicron BA.4/5) Coronavirus (COVID-19) update: FDA authorizes second booster dose of two COVID-19 vaccines for older and immunocompromised individuals. Three doses of an mRNA Covid-19 vaccine in solid-organ transplant recipients. Kamar N, Abravanel F, Marion O, Couat C, Izopet J, Del Bello A. Possible side effects after getting a COVID-19 vaccine. Janssen COVID-19 vaccine.Ĭenters for Disease Control and Prevention. Safety monitoring of an additional dose of COVID-19 vaccine - United States, August 12–September 19, 2021. CDC recommends additional boosters for certain individuals. CDC endorses ACIP’s updated COVID-19 vaccine recommendations.Ĭenters for Disease Control and Prevention. COVID-19 vaccine boosters.Ĭenters for Disease Control and Prevention. Coronavirus (COVID-19) update: FDA takes multiple actions to expand use of Pfizer-BioNTech COVID-19 vaccine.Ĭenters for Disease Control and Prevention. Coronavirus (COVID-19) update: FDA expands eligibility for Pfizer-BioNTech COVID-19 vaccine booster dose to children 5 through 11 years.įood and Drug Administration. Coronavirus (COVID-19) update: FDA expands eligibility for Pfizer-BioNTech COVID-19 booster dose to 16- and 17-year-olds.įood and Drug Administration. COVID-19 vaccines for specific groups of people.įood and Drug Administration. Coronavirus (COVID-19) update: FDA takes additional actions on the use of a booster dose for COVID-19 vaccines.Ĭenters for Disease Control and Prevention. Coronavirus (COVID-19) update: FDA expands eligibility for COVID-19 vaccine boosters.įood and Drug Administration. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
